 The $\mathrm pK_\mathrm a$ of the amino group is not relevant. When would someone calculate an isoelectric point for a compound Whenever a compound has at least one pKa value. The isoelectric point is defined as the pH at which a particular amino acid sequence (peptide chain) bears no net electrical charge. Since the $\mathrm$ is simply the average of the two $\mathrm pK_\mathrm a$ values of the two carboxylic acid groups. Zeta potential is a charge on a given particle that is present at a shear plane. I've searched a lot, and the most helpful post that I found was How do I calculate the isoelectric point of amino acids with more than two pKa's? The isoelectric points can also be measured with the help of zeta potential. I was asked to calculate its isoelectric point, with the given $\mathrm pK_\mathrm a$ values.  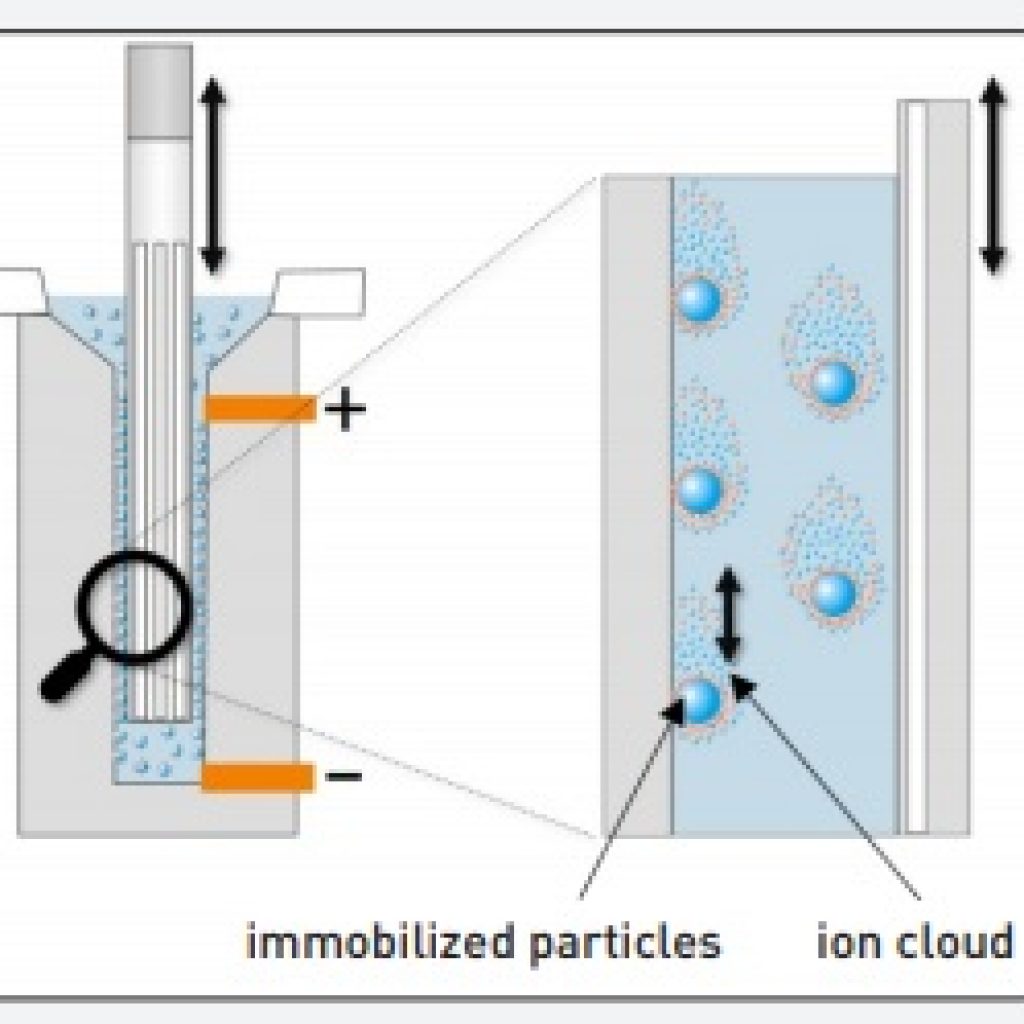
73 strong arguments for performing them at 25C. For this we should refer to the titration curve of the amino acid. As regards the subsequent pH measurements, however, there are. 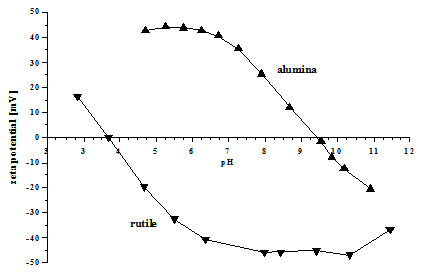
Your confusion seems to stem from choosing the relevant p K a values. It can be calculated by the average of the relevant p K a values as you have mentioned. This movement guarantees that the sample stays homogeneous. Isoelectric point of an amino acid is the p H at which the molecule carries no net charge 1. The standard nomenclature to represent the isoelectric point is pH(I).1 However, pI is also used.2 For brevity, this article uses pI. Each charged particle will migrate in a pattern determined by its particular property due to changes in speed and direction, allowing for the separation of biomolecule components with similar properties.The following amino acid is called lysine. 10 ml of a suspension are constantly agitated during measurement. The isoelectric point(pI, pH(I), IEP), is the pHat which a moleculecarries no net electrical chargeor is electrically neutral in the statistical mean. Negatively charged particles such as nucleic acids gravitate toward the anode, while the positively charged particles toward the cathode. Depending on the mass and net charge of each particle in the solution, ionized biomolecules will migrate at different rates when exposed to an electric field. When charged molecules are placed in an electric field, they travel in the opposite direction of the positive or negative pole. This next zero charge or isoelectric point comes from the average of the 2 pKa. The pI, or isoelectric point, corresponding to the zwitterion form lets you calculate the pH at which an amino acid will have a net zero charge. Use Protein Isoelectric Point when you want. A solution containing biomolecules will have either positively or negatively charged ions depending on the pH. In Conclusion: Amino acids can exist as zwitterions containing a protonated amine group and deprotonated carboxyl group. Protein Isoelectric Point calculates the theoretical pI (isoelectric point) for the protein sequence you enter. Principle of Gel Electrophoresis The principle behind electrophoresis is the observation that the majority of biomolecules exist as electrically charged particles with ionizable functional groups. Gel electrophoresis is one of the laboratory methods for separating DNA, RNA, or protein molecules based on their electric charge or size. A flat-line QRS with no discernible features. This can be either: A biphasic QRS where R wave height Q or S wave depth. In 1948, Arne Tisselius received Novel Prize in Chemistry for his contributions to the electrophoretic technique. The isoelectric (equiphasic) lead is the frontal lead with zero net amplitude. Novex IEF Gels effectively create a pH gradient so proteins separate according to their unique pI. The pI is the pH at which a protein has no net charge and does not move in an electric field. In the 1930s, Swedish biophysicist Arne Tisselius developed electrophoresis while researching blood proteins. Isoelectric focusing (IEF) is an electrophoresis technique that separates proteins based on their isoelectric point (pI). Capillary isoelectric focusing (cIEF) is a high-resolution analytical technique that allows the separation of protein/peptide mixtures, protein glycoforms and. Electrophoresis is a chemical process in which an electric charge in a solution flow toward an opposing electrode. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
